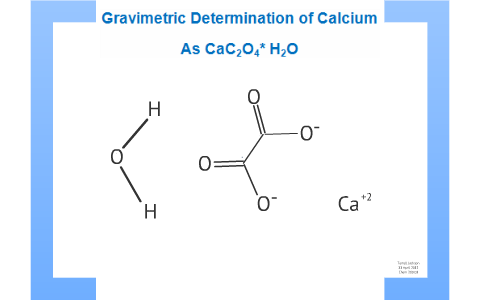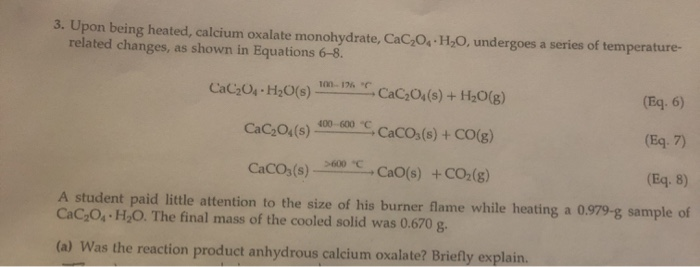
Whewellite, CaC2O4⋅H2O: structural study by a combined NMR, crystallography and modelling approach - CrystEngComm (RSC Publishing)
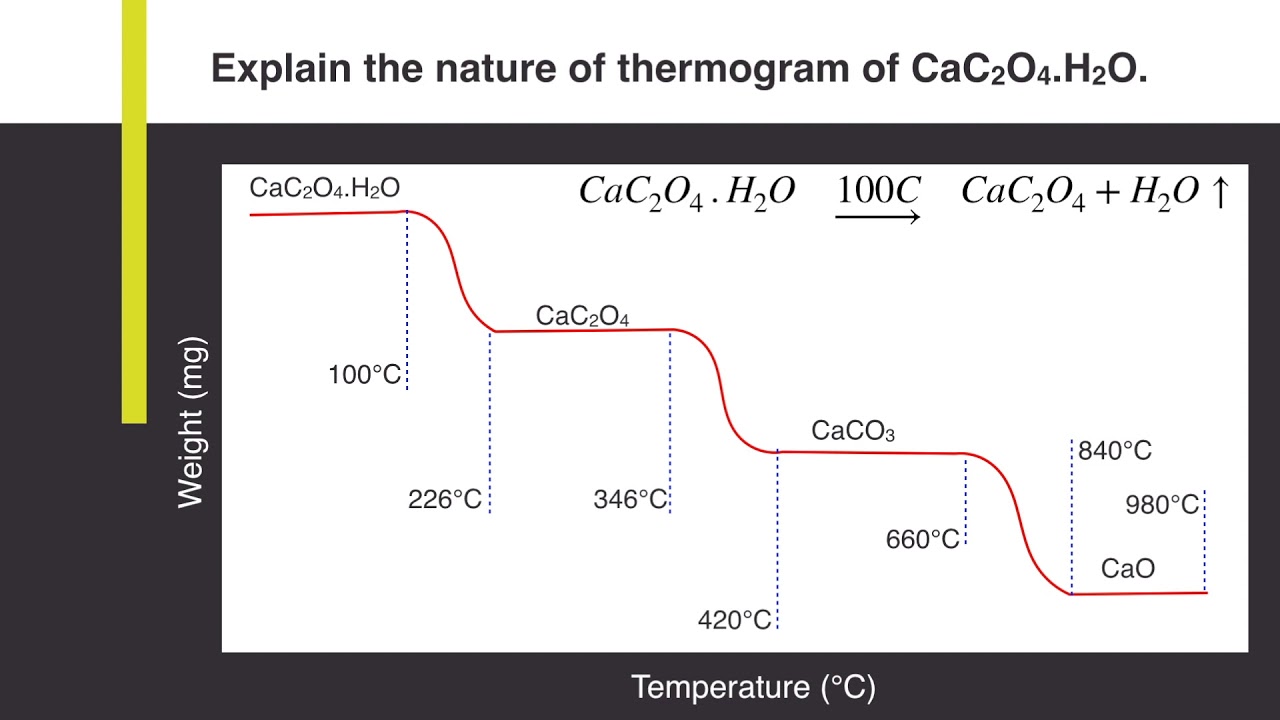
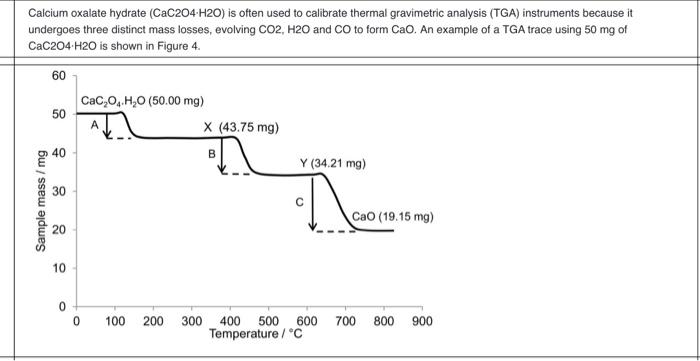
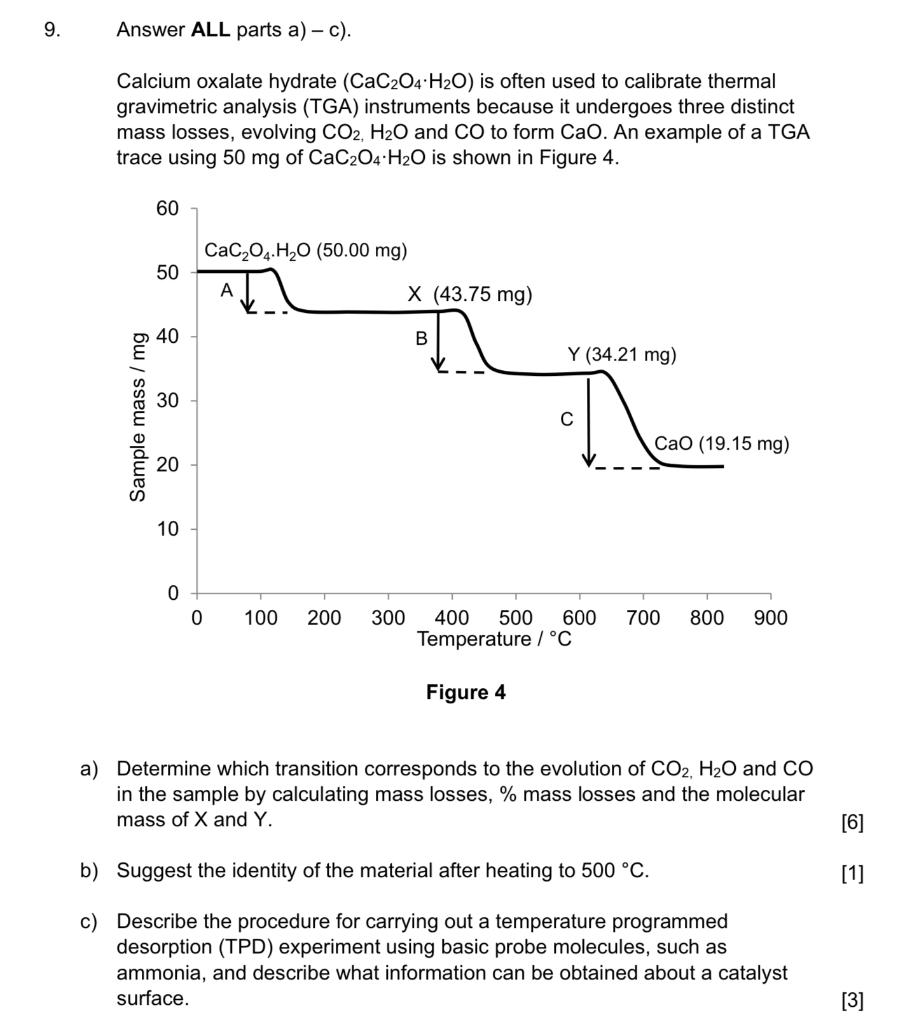
Explain the nature of thermogram of Calcium Oxalate Monohydrate (CaC2O4.H2O) | Analytical Chemistry - YouTube

SOLVED: 3. Upon being heated, calcium oxalate monohydrate, CaC2O4 H2O, undergoes a series of temperature-related changes, as shown in Equations 6-8. CaC2O4 H2O(s) -> CaC2O4(s) + H2O(g) (Eq. 6) CaC2O4(s) + H2O(g) ->

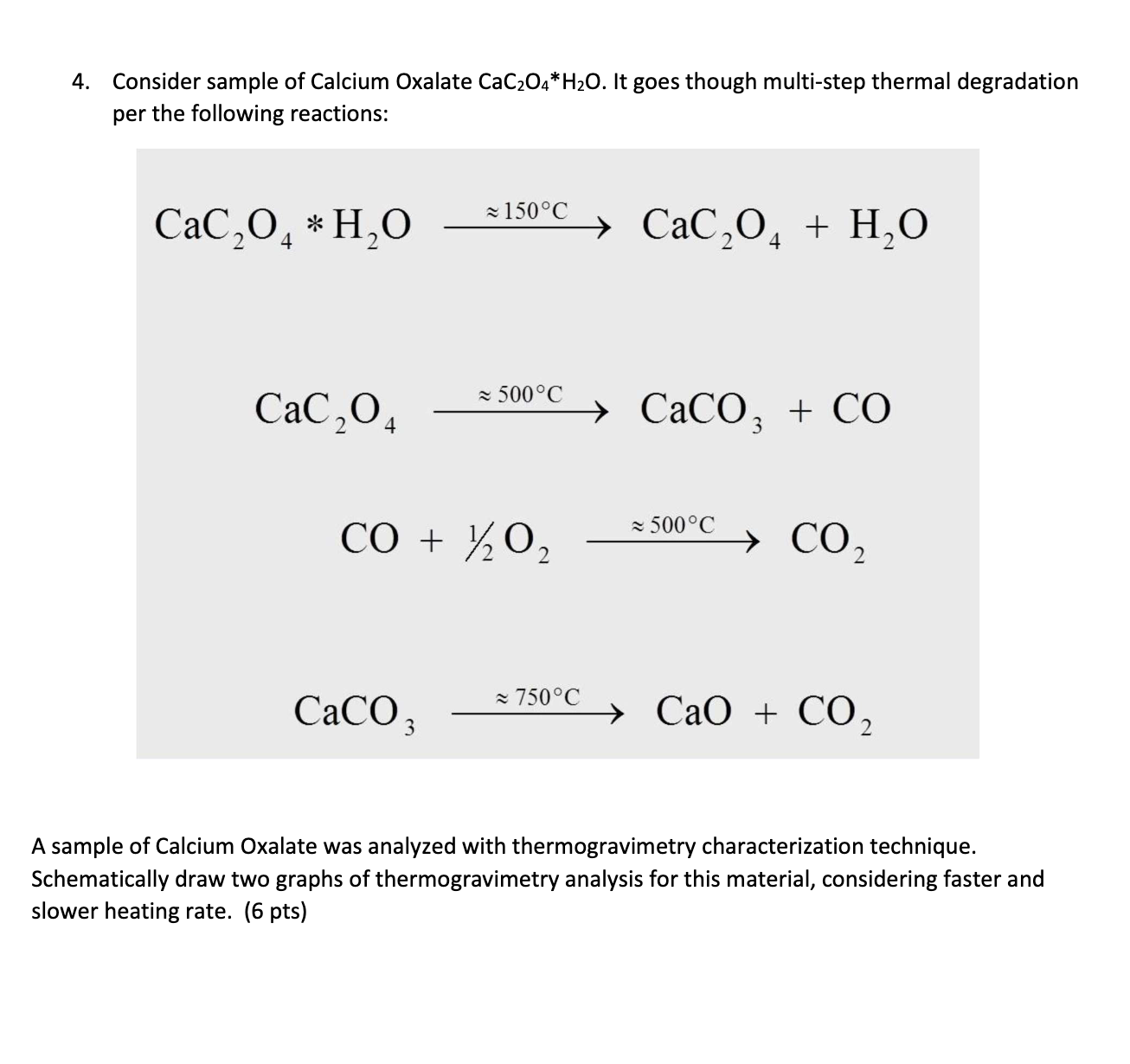
Thermal Decomposition of CaC2O4·H2O Studied by Thermo-Raman Spectroscopy with TGA/DTA | Analytical Chemistry
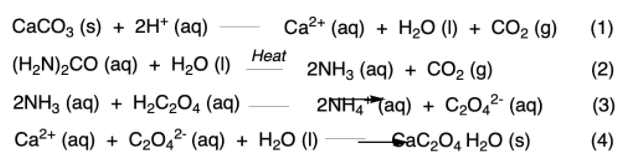
Calcium oxalate, CaC2O4.H2O, is a sparingly soluble salt of analytical and physiological importance. - Sarthaks eConnect | Largest Online Education Community

Thermal Decomposition of CaC2O4·H2O Studied by Thermo-Raman Spectroscopy with TGA/DTA | Analytical Chemistry
MSE310 Lecture 2 Thermogravimetric Analysis (TGA) The TGA technique measures the mass of a sample as it is heated, cooled or he
Calcium oxalate, CaC2O4.H2O, is a sparingly soluble salt of analytical and physiological importance. - Sarthaks eConnect | Largest Online Education Community