Acetic acid, sodium salt, hydrate (1:1:3) (CAS 6131-90-4) - Chemical & Physical Properties by Cheméo

SOLVED: Solution Balanced Net Ionic Equation: 1. Sodium acetate and water: NaCH3COO(aq) + H2O(l) -> CH3COOH(aq) + NaOH(aq) 2. Acetic acid and water: CH3COOH(aq) + H2O(l) -> CH3COOH(aq) + H2O(l) 3. Sodium

SOLVED: Sodium acetate (NaC2H3O2) is a basic salt. When sodium acetate is dissolved in water, it dissociates into its component ions. This reaction goes to completion, as indicated by the one-way arrow
Acetic acid, sodium salt, hydrate (1:1:3) (CAS 6131-90-4) - Chemical & Physical Properties by Cheméo

Sodium Acetate(CH3COONa) - Structure, Properties, Preparations, Uses, Important questions, FAQs of sodium acetate.

Sodium acetate hydrate, Puratronic™, 99.9985% (metals basis), Thermo Scientific Chemicals | Fisher Scientific
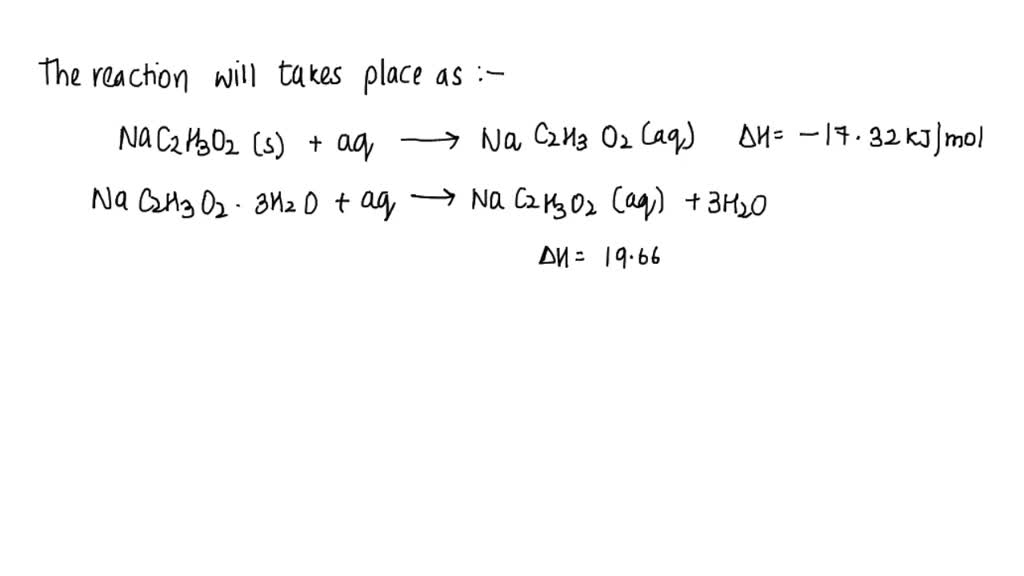
SOLVED: How do you calculate the enthalpy of hydration from the enthalpy of solution? My two salts are anhydrous sodium acetate (C2H3NaO2) and sodium acetate trihydrate (NaC2H3O2·3H2O). The enthalpy of solution for

Jual Sodium Acetate C2H3Nao2 Garam Natrium Teknis 500 Gram - Jakarta Barat - Jauharah-store | Tokopedia







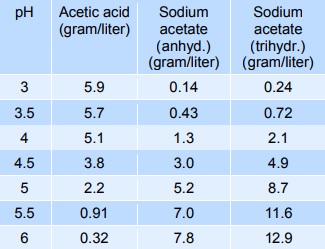





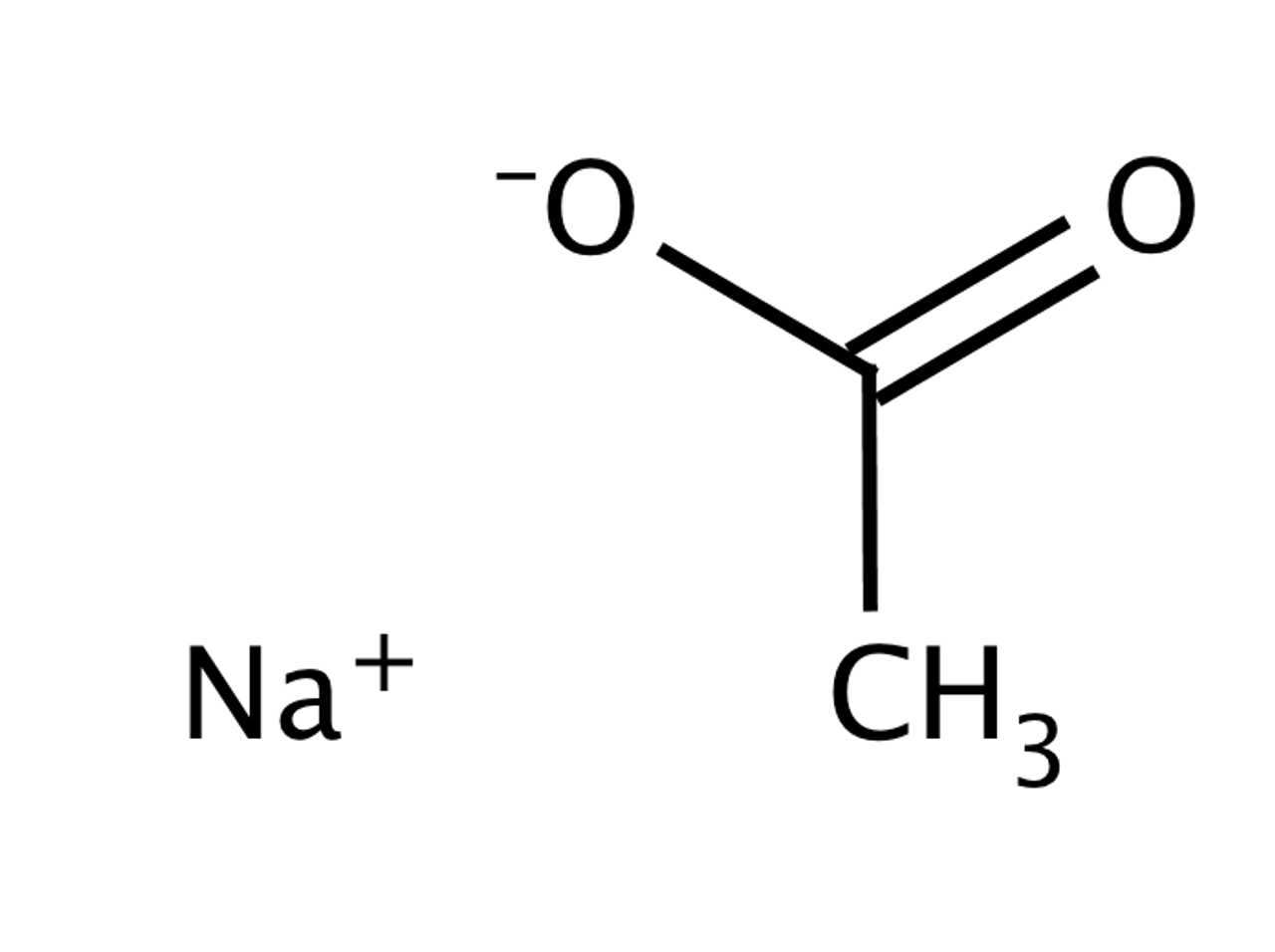
![Sodium acetate - Optional[17O NMR] - Chemical Shifts - SpectraBase Sodium acetate - Optional[17O NMR] - Chemical Shifts - SpectraBase](https://spectrabase.com/api/spectrum/BnljDgyGm6a/structure.png?h=300&w=382)