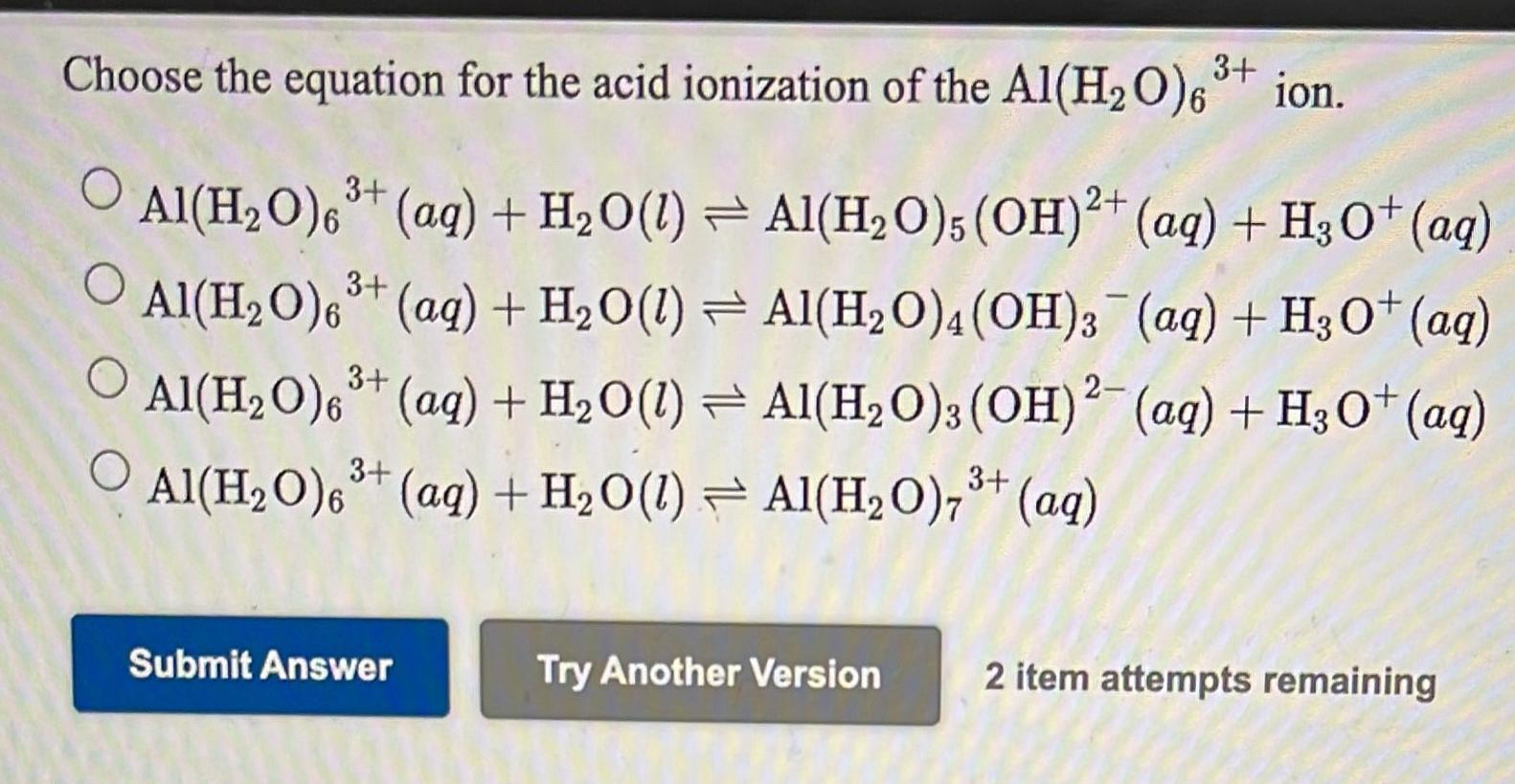
![Q.52 In the following [Al(H2O).)] + HCO3- [Al(H2O),OH)2 + HCO, (b) species behaving as Bronsted - Lowry acids are: Q.52 In the following [Al(H2O).)] + HCO3- [Al(H2O),OH)2 + HCO, (b) species behaving as Bronsted - Lowry acids are:](https://toppr-doubts-media.s3.amazonaws.com/images/2164712/a42abbba-24aa-49e2-9c74-16ac3879c732.jpg)
Q.52 In the following [Al(H2O).)] + HCO3- [Al(H2O),OH)2 + HCO, (b) species behaving as Bronsted - Lowry acids are:
NaOH + PbO = x + H2O NaOH + SnO2 = y + H2O NaOH + H2O + Al = z + H2 Sum of number of atom present in one molecule
![SOLVED: In Al3+ + 6H2O â†' [Al(H2O)6]3+, what does H2O act as? Lewis base, Bronsted-Lowry base, Bronsted-Lowry acid, Lewis Acid 2. Correctedtext: What will happen if you remove a reactant from a SOLVED: In Al3+ + 6H2O â†' [Al(H2O)6]3+, what does H2O act as? Lewis base, Bronsted-Lowry base, Bronsted-Lowry acid, Lewis Acid 2. Correctedtext: What will happen if you remove a reactant from a](https://cdn.numerade.com/ask_previews/89fd73fd-55ff-4e1c-b8f2-49c556eb68c1_large.jpg)
SOLVED: In Al3+ + 6H2O â†' [Al(H2O)6]3+, what does H2O act as? Lewis base, Bronsted-Lowry base, Bronsted-Lowry acid, Lewis Acid 2. Correctedtext: What will happen if you remove a reactant from a
Al(H2O)6 3+ + H2O structures from MD simulations, all with d(Al-OW′)... | Download Scientific Diagram
The aluminium solution which contains [Al(H2O) 6] ^(3+) ions have the tendency to hydrolyze. Why? - Quora

SOLVED: 1 Balance en la ecuación químicas Al2O3 + H2O -> AL(OH)3 Por favor 2071 u 2 0 7H + b) 4 ecvació € ( H0)| V U 0 ví m'ca

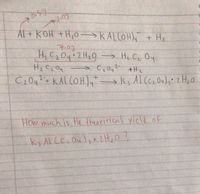










![What is the conjugate base of [Al (H2O)3 (OH)3]- ? Give the reason be - askIITians What is the conjugate base of [Al (H2O)3 (OH)3]- ? Give the reason be - askIITians](https://files.askiitians.com/cdn1/question-images/253949-screenshot_2019-08-19-13-20-17.png)