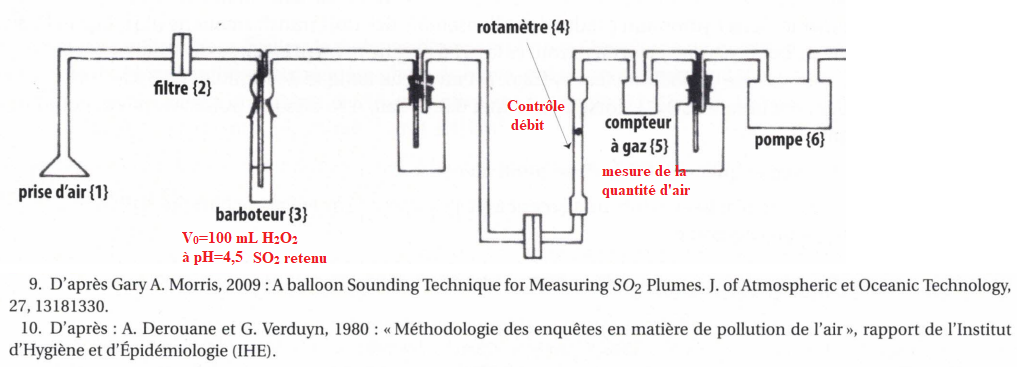
Crystals | Free Full-Text | Phase Formation in NaH2PO4–VOSO4–NaF–H2O System and Rapid Synthesis of Na3V2O2x(PO4)2F3-2x
![Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram](https://www.researchgate.net/publication/286004505/figure/fig3/AS:1132193107722246@1646947280502/Angular-variation-of-EPR-line-positions-of-VO-ZAPH-in-the-plane-a-ab-b-bc-and-c_Q320.jpg)
Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram
Theoretical Study of Oxovanadium(IV) Complexation with Formamidoximate: Implications for the Design of Uranyl-Selective Adsorben
![Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram](https://www.researchgate.net/publication/286004505/figure/fig4/AS:1132193107722247@1646947280535/Angular-variation-in-the-three-planes-ab-bc-and-ca-in-VOZAPH-for-the-quantities-a-g_Q320.jpg)
Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram
A Coordination Chemistry Study of Hydrated and Solvated Cationic Vanadium Ions in Oxidation States +III, +IV, and +V in Solution
A Coordination Chemistry Study of Hydrated and Solvated Cationic Vanadium Ions in Oxidation States +III, +IV, and +V in Solution
![Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram](https://www.researchgate.net/publication/286004505/figure/fig6/AS:1132193107722252@1646947280620/Orientations-of-the-VO-ions-and-VOH2O5-complexes-in-the-ZAPH-lattice-a.jpg)
Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram
![Molecular formulas of (1) [VO(dipic)(H2O)2]•2 H2O, (2) [VO(dipic)(bipy... | Download Scientific Diagram Molecular formulas of (1) [VO(dipic)(H2O)2]•2 H2O, (2) [VO(dipic)(bipy... | Download Scientific Diagram](https://www.researchgate.net/publication/361558060/figure/fig1/AS:1175627562532865@1657302861975/Molecular-formulas-of-1-VOdipicH2O22-H2O-2-VOdipicbipy-VOidabipy2.png)
Molecular formulas of (1) [VO(dipic)(H2O)2]•2 H2O, (2) [VO(dipic)(bipy... | Download Scientific Diagram
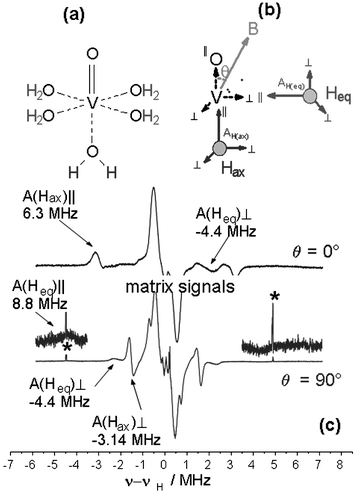
Principles and applications of ENDOR spectroscopy for structure determination in solution and disordered matrices - Chemical Society Reviews (RSC Publishing) DOI:10.1039/B500509B

Figure 2 from Crystal growth of four oxovanadium(IV) tartrates prepared via a mild two-step hydrothermal method: observation of spin-dimer behavior and second harmonic generation. | Semantic Scholar

A Coordination Chemistry Study of Hydrated and Solvated Cationic Vanadium Ions in Oxidation States +III, +IV, and +V in Solution and Solid State | Inorganic Chemistry
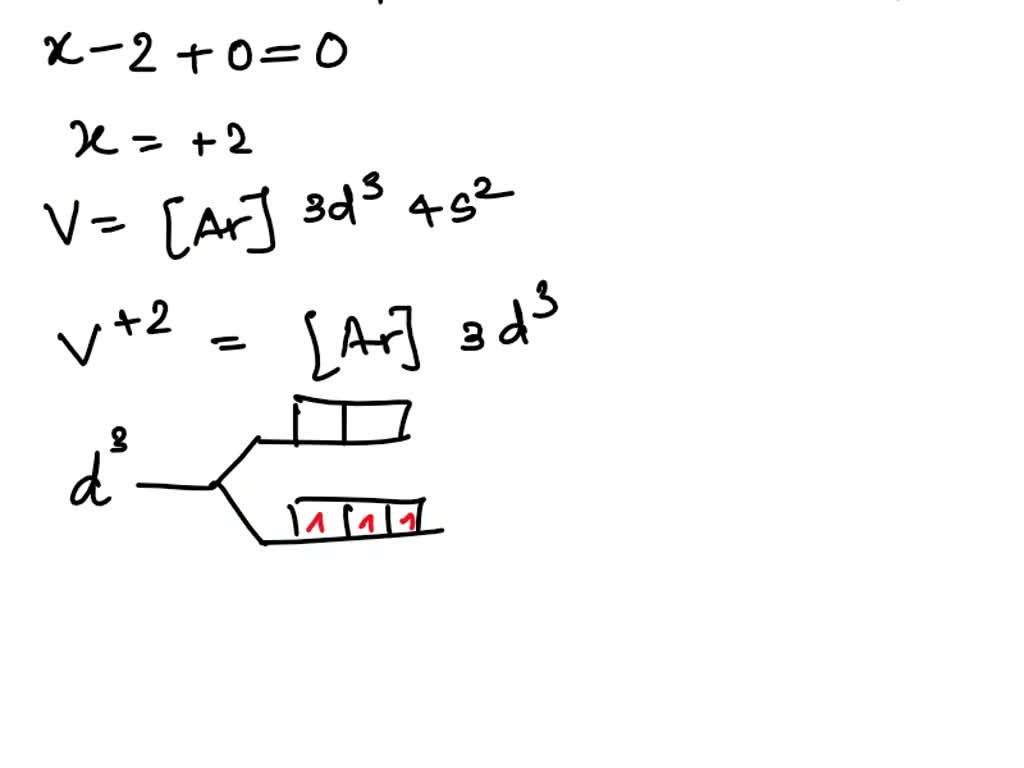
SOLVED: How many unpaired electrons is VO(H2O)5 expected to have and how could it be proven experimentally?

Transition Metals Occupy the d-block of periodic table Have d-electrons in valence shell Some characteristics of Transition Metals and their compounds. - ppt download
2016 Ch112 – problem set 5 Due: Thursday, Nov. 7 Remember your project report is due next week (Nov. 22) 1 Problem 1 (2 points
![Vanadium transition metal Chemistry ions vanadium(II) V2+ vanadium(III) V3+ vanadium(IV) [VO2]2+ vanadium(V) VO2+ vanadate VO43- redox chemical reactions complexes demonstration of oxidation states principal oxidation states +2 +3 +4 +5 GCE AS Vanadium transition metal Chemistry ions vanadium(II) V2+ vanadium(III) V3+ vanadium(IV) [VO2]2+ vanadium(V) VO2+ vanadate VO43- redox chemical reactions complexes demonstration of oxidation states principal oxidation states +2 +3 +4 +5 GCE AS](https://www.docbrown.info/page07/SSquestions/0complexVH2O6b.gif)
Vanadium transition metal Chemistry ions vanadium(II) V2+ vanadium(III) V3+ vanadium(IV) [VO2]2+ vanadium(V) VO2+ vanadate VO43- redox chemical reactions complexes demonstration of oxidation states principal oxidation states +2 +3 +4 +5 GCE AS
![Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram](https://www.researchgate.net/publication/286004505/figure/fig2/AS:1132193107722244@1646947280476/EPR-spectrum-of-a-VO-doped-ZAPH-single-crystal-with-the-magnetic-field-at-0-from-the_Q320.jpg)
Orientations of the VO²⁺ ions and [VO(H2O)5]²⁺ complexes in the ZAPH... | Download Scientific Diagram

Insights on the mechanistic features of catalytic oxidations of simple and conjugated olefins promoted by VO(acac)2/H2O2 system, in acetonitrile: A computational study - ScienceDirect
![SOLVED: B – Complexes Complex Ion Coordination Number Molecular Geometry Name [VO2(H2O)3]+ [VO(H2O)5]2+ [V(H2O)6]3+ Analysis 1. Write a balanced redox equation for each conversion using Zn as the reducing agent. a) VO2+ SOLVED: B – Complexes Complex Ion Coordination Number Molecular Geometry Name [VO2(H2O)3]+ [VO(H2O)5]2+ [V(H2O)6]3+ Analysis 1. Write a balanced redox equation for each conversion using Zn as the reducing agent. a) VO2+](https://cdn.numerade.com/ask_previews/c07def3e-6d56-40ff-a5e3-203dd65cb17f.gif)


![ESR spectrum of [VO(L)(H2O)2].2H2O 3. | Download Scientific Diagram ESR spectrum of [VO(L)(H2O)2].2H2O 3. | Download Scientific Diagram](https://www.researchgate.net/publication/371950179/figure/fig3/AS:11431281176496360@1690189391233/ESR-spectrum-of-VOLH2O22H2O-3.png)