In situ ATR-FTIR study of H2O and D2O adsorption on TiO2 under UV irradiation - Physical Chemistry Chemical Physics (RSC Publishing)
![PDF] The adsorption of H2O on TiO2 and SnO2(110) studied by first-principles calculations | Semantic Scholar PDF] The adsorption of H2O on TiO2 and SnO2(110) studied by first-principles calculations | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/927280f2bedb1fb518ae18454a490f3c9ef5ccaa/6-Table1-1.png)
PDF] The adsorption of H2O on TiO2 and SnO2(110) studied by first-principles calculations | Semantic Scholar

SOLVED: The reaction of 1.23 g TiCl4 (FW 189.71) with excess H2O according to the following reaction yields 0.456 g of TiO2 (FW 79.87). What is the percent yield? TiCl4 + 2
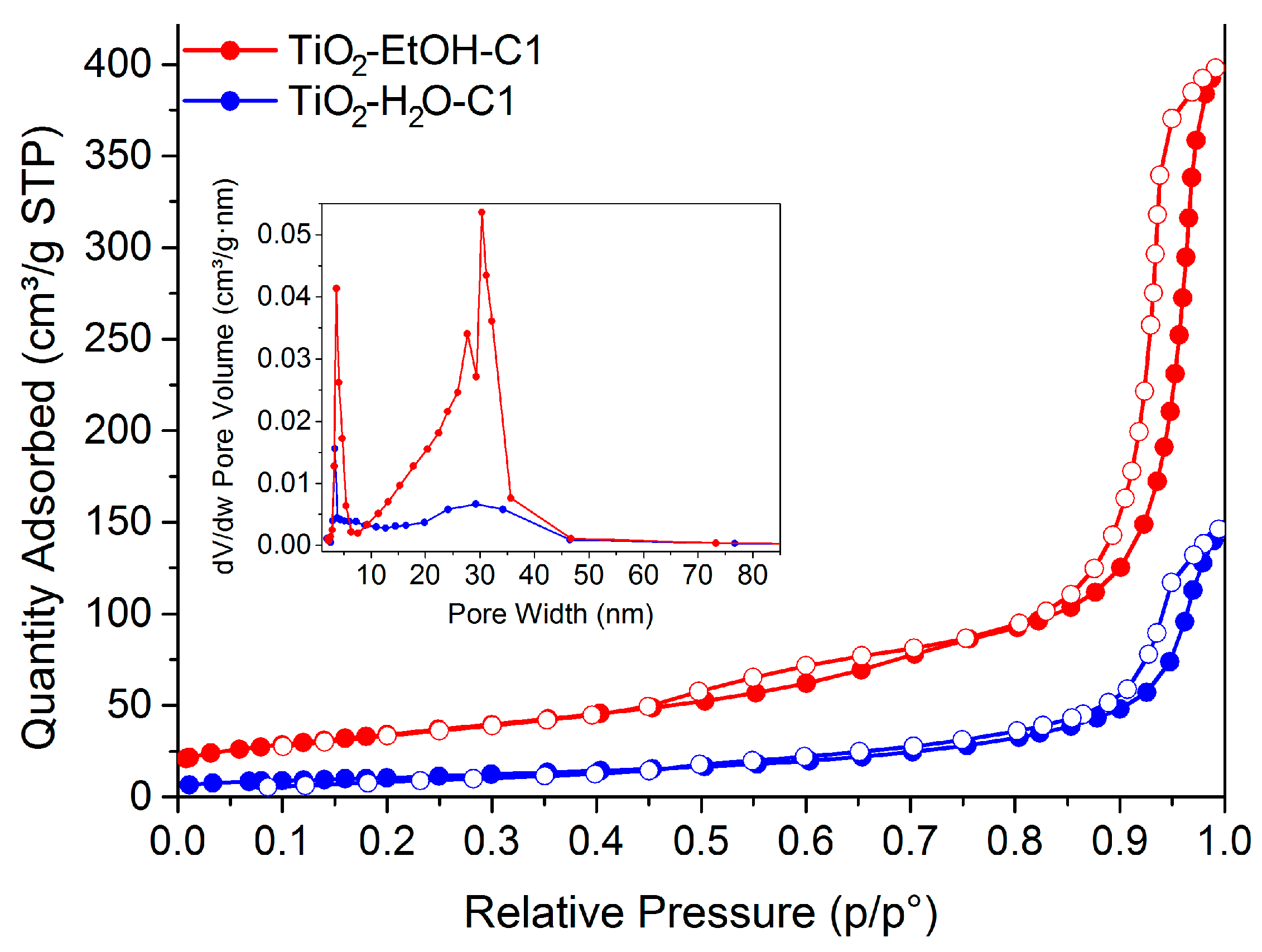
Nanomaterials | Free Full-Text | Novel Sol-Gel Synthesis of TiO2 Spherical Porous Nanoparticles Assemblies with Photocatalytic Activity

Hydrogen-Bond Network Promotes Water Splitting on the TiO2 Surface | Journal of the American Chemical Society

Full article: Effectively inhibiting particles aggregation and sedimentation for TiO2-H2O suspension by application of an electrode

TiO2 Phase Engineering by Millisecond Range Annealing for Highly Efficient Photocatalysis | The Journal of Physical Chemistry C

Water adsorption on TiO2 surfaces probed by soft X-ray spectroscopies: bulk materials vs. isolated nanoparticles | Scientific Reports

Direct Four‐Electron Reduction of O2 to H2O on TiO2 Surfaces by Pendant Proton Relay - Sheng - 2013 - Angewandte Chemie International Edition - Wiley Online Library
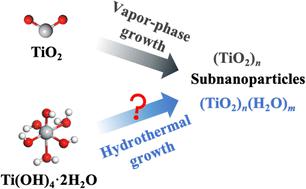
Aqueous growth of titania subnanoparticles: an understanding of the ultrasmall visible-light-absorbing unit of (TiO2)8(H2O)16,Physical Chemistry Chemical Physics - X-MOL