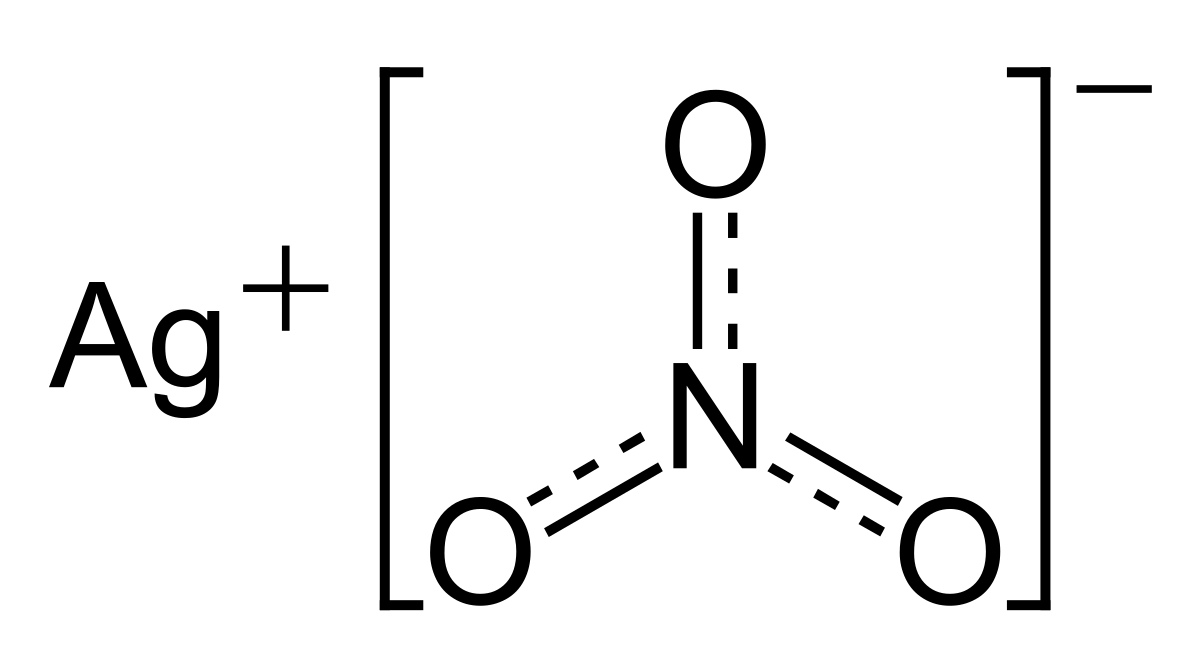
Intermolecular Oxidative Radical Addition to Aromatic Aldehydes: Direct Access to 1,4- and 1,5-Diketones via Silver-Catalyzed Ring-Opening Acylation of Cyclopropanols and Cyclobutanols | The Journal of Organic Chemistry

Balance the following chemical reaction: 1. AgNO3 - Ag + NO2 + O2 2. ZnS + O2 ZnO + SO2 3. S + H2SO4 - SO2 + H2O 4. Zn + KOH K2ZnO2 + H2 5. NO2 + H2O → HNO3 + HNO2

OneClass: Hess's Law (b) Given the following thermochemical data: Ag(s)+HNO3(aq)â†'AgNO3(aq)+½H2(g) ...
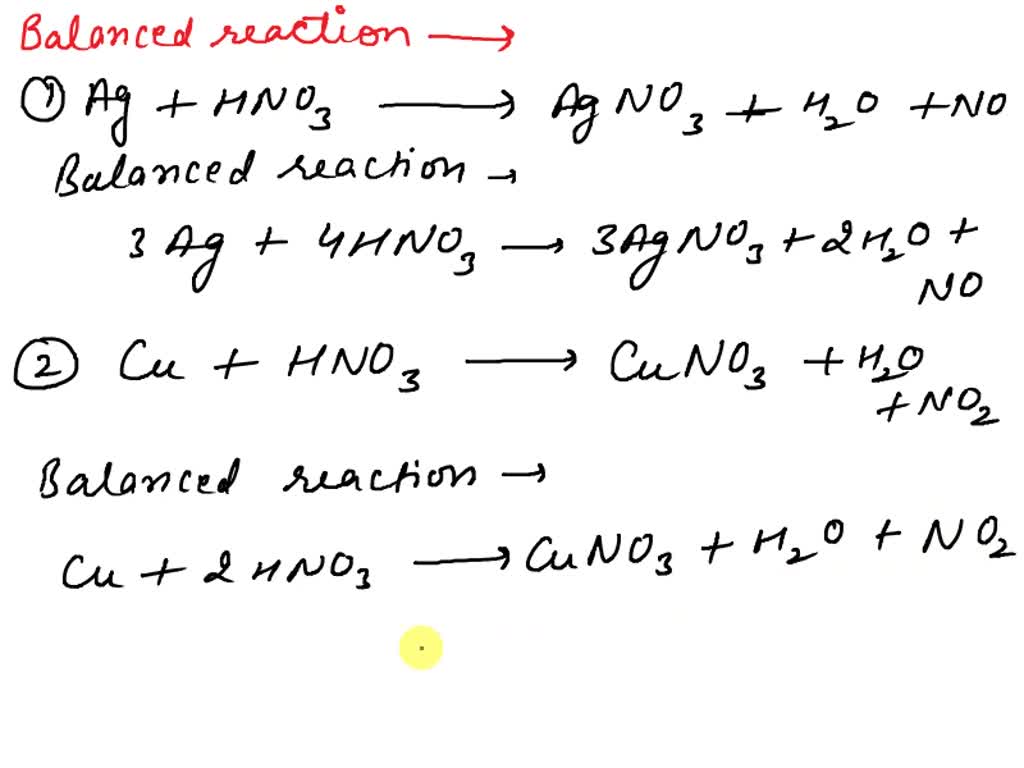
SOLVED: Balance the following reactions involving metal nitrates: Ag + HNO3 â†' AgNO3 + H2O + NO Cu + HNO3 â†' Cu(NO3)2 + H2O + NO AgNO3 + Cu â†' Cu(NO3)2 +
![The volume (in mL) of 0.1 M AgNO3 required for complete precipitation of chloride ions present in 30 mLof 0.01 M solution of [Cr(H2O)5Cl]Cl2, as silver chloride is close toCorrect answer is ' The volume (in mL) of 0.1 M AgNO3 required for complete precipitation of chloride ions present in 30 mLof 0.01 M solution of [Cr(H2O)5Cl]Cl2, as silver chloride is close toCorrect answer is '](https://edurev.gumlet.io/ApplicationImages/Temp/24d955be-fbfb-4c5b-9701-95bc154f720c_lg.jpg)