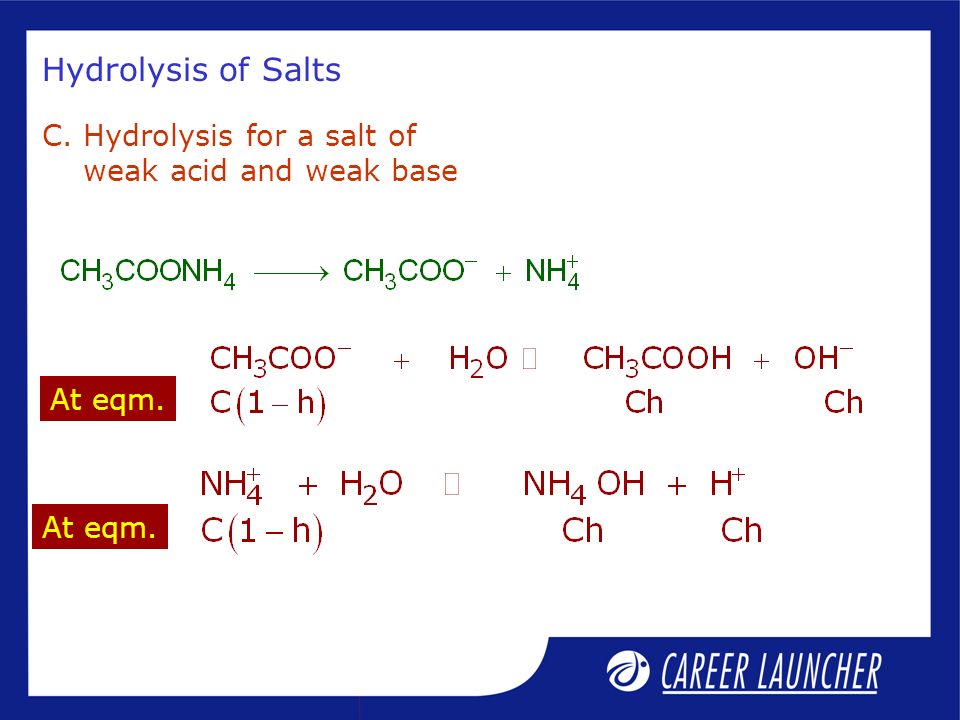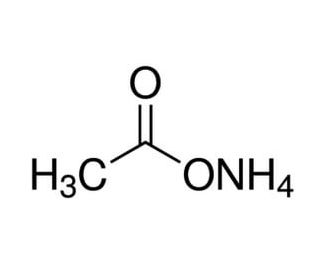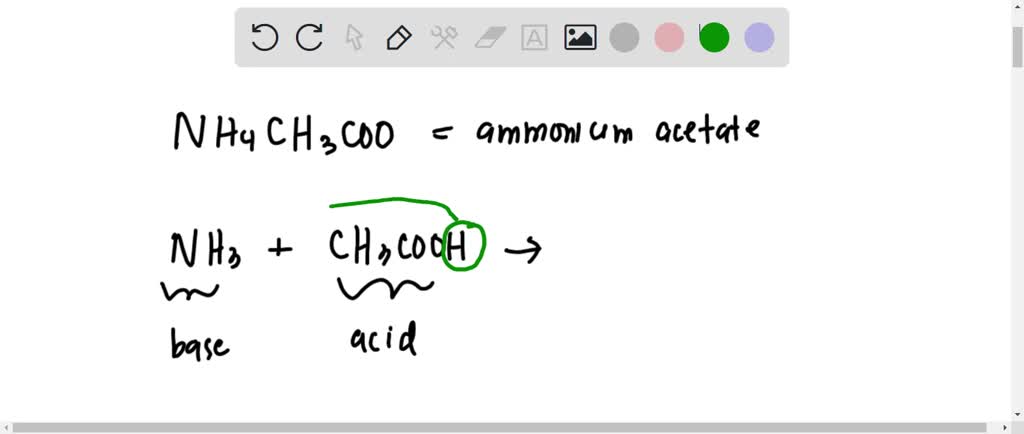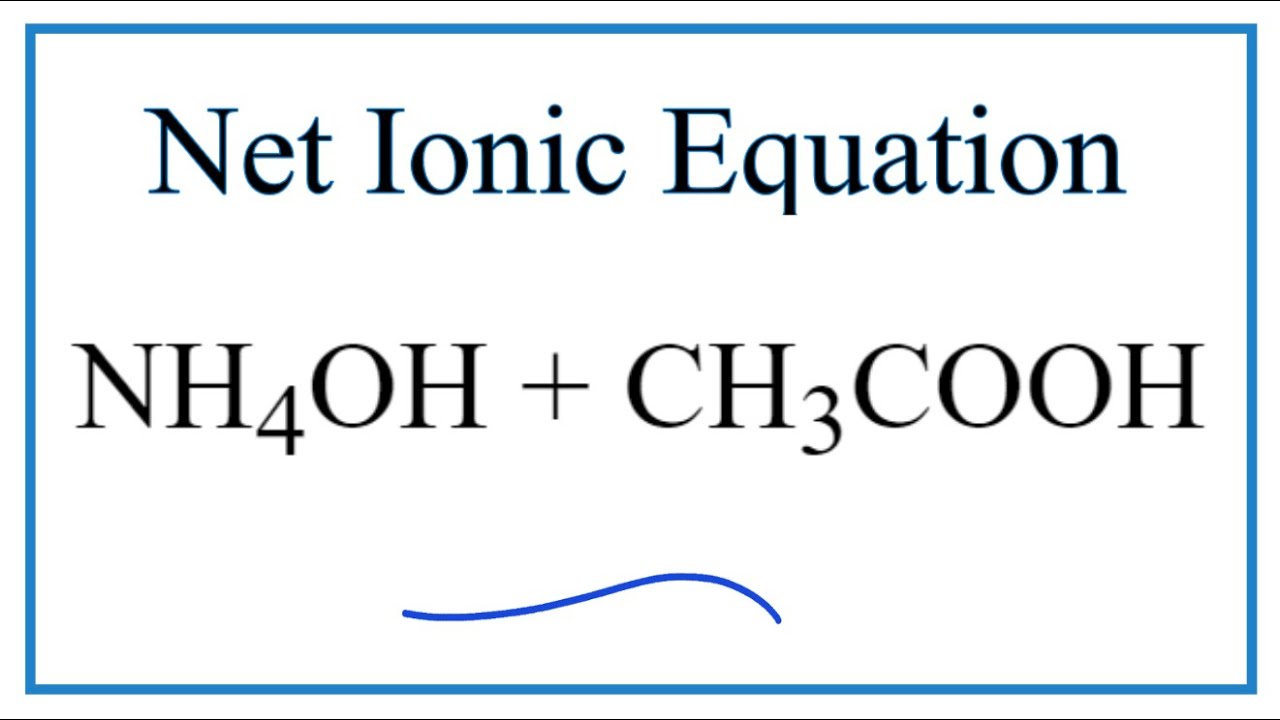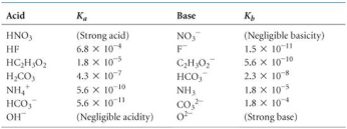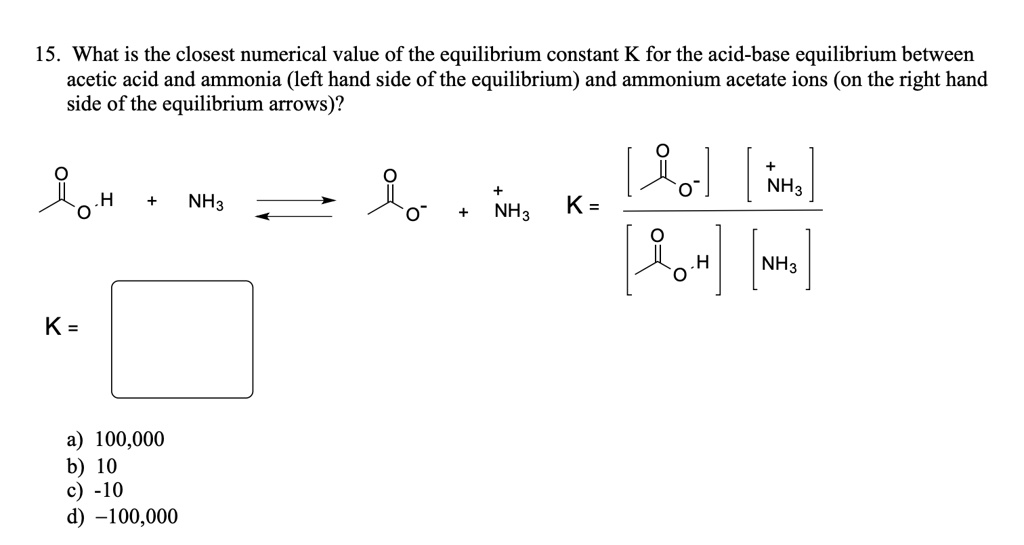
SOLVED: 15. What is the closest numerical value of the equilibrium constant K for the acid-base equilibrium between acetic acid and ammonia (left hand side of the equilibrium) and ammonium acetate ions (

3. Salt of a of a weak acid and a weak base both the car Maximum hydrolysis the cation and anion ar and OH - roduce H ut it can be either

Formation of adenine from CH3COONH4/NH4HCO3—the probable prebiotic route for adenine - ScienceDirect

Green Synthesis of 1,4‐Dihydropyridine Derivative in Water - Isomura - 2018 - ChemistrySelect - Wiley Online Library
![Welcome to Chem Zipper.com......: [3] CATIONIC AS WELL AS ANIONIC HYDROLYSIS: Welcome to Chem Zipper.com......: [3] CATIONIC AS WELL AS ANIONIC HYDROLYSIS:](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEgL8SQhnjKci8Sd_t_-hwKS6TbUzdVEN36ZVX7jzyaaImcB0R3-pjiQk5I7lhygJIO2mioVSY0269QkoVgPgOTPfaPDfCu_McwLwOKS5MWlvzzjd4qRRjyHV5vZto0d67HeHHMAvK9k81o/s1600/SH28.PNG)