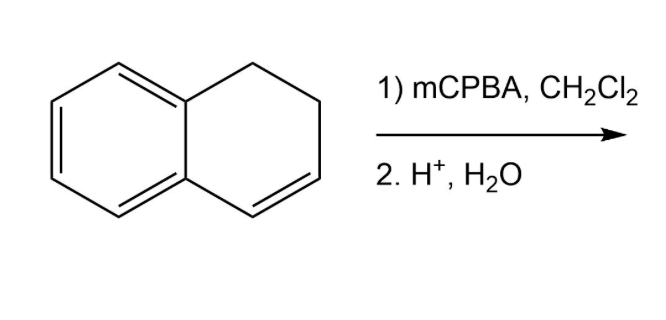
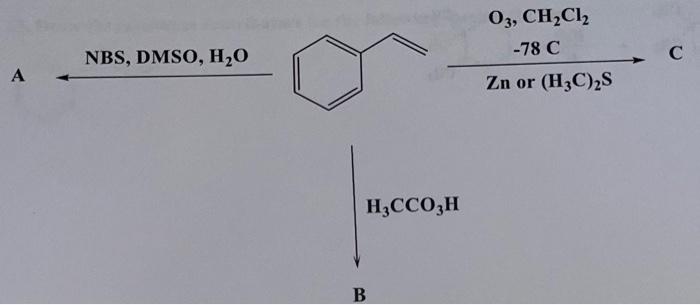
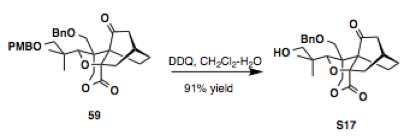
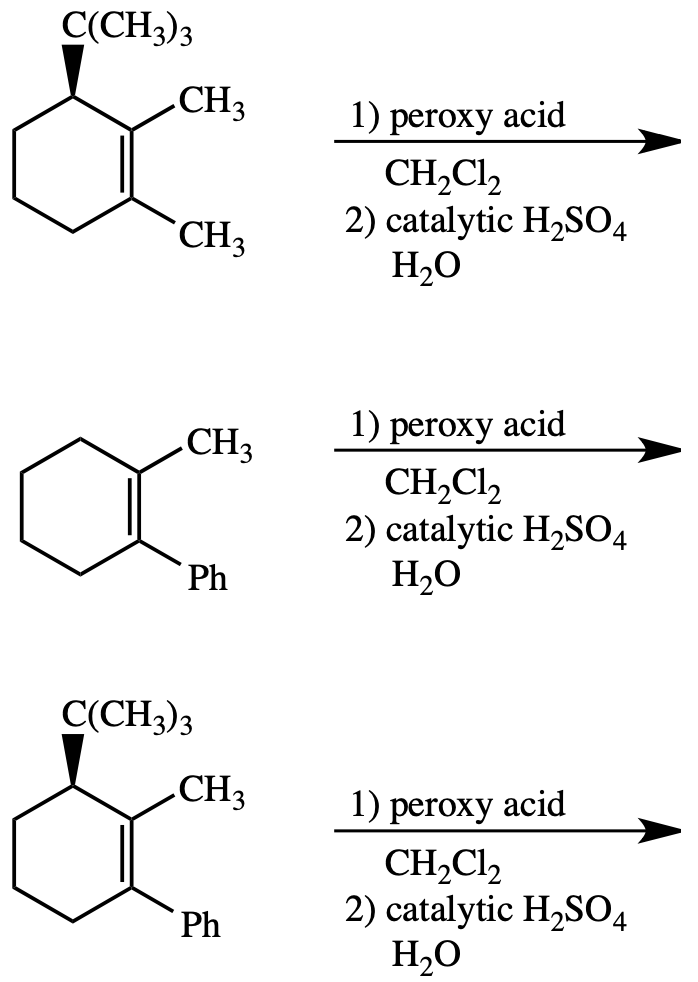
Synthesis of (²H)-, (¹³C)-, and (¹³C,¹⁸O)-4. (a) PCC, CH2Cl2, rt; (b)... | Download Scientific Diagram

SOLVED: Water (H2O) and dichloromethane (CH2CI2) are common solvents in chemistry lab. Which of these solvents is expected to have a higher boiling point? Select one: Water because the molecules in water

SEM images obtained with each investigated monomer in CH2Cl2 + H2O sat.... | Download Scientific Diagram

SOLVED: Water (H2O) and dichloromethane (CH2Cl2) are common solvents in a chemistry lab. Which of these solvents is expected to have a higher boiling point? Select one: a. Water because the molecules

Cyclic voltammogram of CH2Cl2 (left) and CH2Cl2 + H2O (right) with 0.1... | Download Scientific Diagram

SOLVED: Water (H2O) and dichloromethane (CH2Cl2) are common solvents in a chemistry lab. Which of these solvents is expected to have a higher boiling point? Select one: a. Water because the molecules

Wettability data for the polymers obtained in CH2Cl2 and CH2Cl2 + H2O,... | Download Scientific Diagram

Wettability data for the polymers obtained in CH2Cl2 and CH2Cl2 + H2O,... | Download Scientific Diagram

SOLVED: When methylene chloride (CH2Cl2) is poured into water (H2O), they do not mix but form two separate layers. Water forms the upper layer and the methylene chloride forms the lower layer.

What's the product when 1,3 butanediol react with a) PCC/CH2Cl2 b)Na2CrO7/H2SO4, H2O and 1-methyl 1,2-cyclohexanediol react with NaOCl/AcOH? | Homework.Study.com